- What is hydrogen peroxide: chemical formula and properties Chemical formula
- Chemical properties
- In traditional medicine
Hydrogen peroxide was first obtained by the chemist Luis Tenero in 1818 and very soon began to be used in industry. Now it is widely used in households and everyday life.
Differences between ammonia and ammonia
The main difference between ammonia and ammonia is their initial state of aggregation. Ammonia is a gas that liquefies at a temperature of -33 degrees Celsius. As for ammonia, this substance is a liquid . In addition, such a product is sometimes called an ammonia solution. The only similarity can be considered an unpleasant ammonia smell.
It should be noted that for household needs, ammonia is obtained by mixing water and ammonia solution. For medical purposes, the process of forming the product is slightly different and consists of 2 steps. First you need to obtain ammonium hydrate, and then make ammonia from it.
Industrial applications of ammonia
The main share of produced technical ammonia is used in the chemical industry:
- for the production of ammonium and ammonium nitrate fertilizers (ammonium and nitrate nitrate, ammonium chloride, ammonium sulfate and others), widely used in agricultural production for growing various crops, as well as in private gardening;
- when producing soda using the ammonia-chloride method, a concentrated saline solution is saturated with ammonia, through which carbon dioxide is passed under pressure;
- synthetic ammonia is used in the production of nitric acid by catalysis;
- for the production of ammonites - ammonia explosives;
- liquid ammonia is used as a solvent for various inorganic and organic substances;
- in refrigeration units, liquid ammonia is used as refrigerant R717;
- as a rocket fuel oxidizer.
Medical products used in everyday life
Ammonia and ammonia are not the only medical products that can be used for household purposes. Housewives often use ammonium chloride and hydrogen peroxide in everyday life.
Not all of the above products can be purchased at pharmacies. For example, ammonia and ammonia are sold only in specialized stores or household chemical departments. It should be noted that the composition, as well as the process for obtaining these drugs, differ from each other. This can be seen by paying attention to the formula of each substance.
Let's understand the chemical formulas
People who do not understand chemistry believe that ammonia, ammonia, ammonia and hydrogen peroxide are substances similar in composition. In fact, everything is not quite like that. Of course, some of the drugs have the same smell, but they differ in the chemical formula, as well as in the method of preparation.
As for ammonia, its appearance resembles a white powder. Ammonia has an external resemblance only to hydrogen peroxide. Although they also differ in their composition.
Ammonia formula
Ammonia is a colorless gas with a pungent odor. Chemically, such a substance is written with the following formula: NH3. The melting point of the gas is -77.73 degrees Celsius. As for the boiling point, it is -33.34 degrees Celsius.
To produce ammonia industrially, specialists combine hydrogen with nitrogen. This can be written with the following formula: 3H2+N2=2NH3.
In laboratory conditions, ammonium is used, which is exposed to strong alkalis.
Ammonia and its formula
Unlike ammonia, ammonia is a colorless liquid with a pungent odor. This substance has the following chemical formula: NH4OH. Sometimes you can find another formula: NH3∙H2O. A similar notation is used for a 10% aqueous ammonia solution. Many people wonder why an aqueous ammonia solution is called alcohol. It's simple, from the Latin word "spiritus" means evaporation. The same applies to ammonia. If the container is left open, the substance will quickly evaporate.
Ammonia formula
Ammonium chloride is obtained naturally and chemically. Its formula is written as follows: NH4Cl.
It is noteworthy that people have been making ammonia since ancient times. Residents of Ancient Greece obtained ammonium chloride by sublimating soot from the chimney of a furnace heated by camel feces.
In nature, the substance can be found inside cracks in the earth's crust and near volcanoes. Typically, ammonium chloride appears as a white residue.
About the basic properties of ammonia
The variety of applications of ammonia is due to its physical and chemical properties. Ammonia (chemical formula NH3) is a colorless gas with a strong, unpleasant odor. With prolonged exposure to the human body, ammonia causes damage to various organs.
Ammonia enters into chemical reactions with other elements and compounds to form new substances. For example, the reaction of anhydrous ammonia with nitric acid produces ammonium nitrate, which is used in the production of explosives and nitrogen fertilizers.
Ammonia is capable of reducing various metals from their oxides.
How are the substances used?
Some people do not know, but substances such as hydrogen peroxide and ammonia, as well as ammonia powder and ammonia can be used both in medicine and for household purposes.
The above substances can be used:
- In the country;
- As furniture cleaners;
- For washing;
- In cosmetology;
- As insect repellents.
The list can be listed endlessly, the main thing is to know the proportions and additional substances to make a useful drug.
How to use ammonia
A substance such as ammonia has become widespread. Most often, this gas is used to produce fertilizers. It is used for industrial purposes, namely:
- As a neutralizer for acidic waste;
- For creating nylon and nylon;
- For cleaning and dyeing cotton, wool and silk;
- As a refrigerant for freezing units.
At home, the substance is useful only for the garden, to prepare fertilizers yourself. You should remember about safety, so it is better to carry out all operations after using the respirator. You can buy ammonia in specialized stores.
How to use aqueous ammonia
Ammonia aqueous or ammonia is used both in medicine and in the household. This remedy can bring an unconscious person back to his senses. It is enough to open a bottle of ammonia, and then moisten a cotton swab, which you just need to bring to the person’s nose.
Sometimes situations arise when it is necessary to induce a gag reflex. To do this, just mix 10 drops of an aqueous ammonia solution and 100 milliliters of water. It is strictly forbidden to swallow undiluted ammonia, otherwise you may get a burn to the esophagus or stomach. It is necessary to use ammonia only in extreme cases and in small quantities.
As for everyday life, aqueous ammonia allows you to clean suede items. To prepare the solution you will need 4 parts clean water and 1 part alcohol. After mixing, you need to moisten a cotton swab and then wipe the contaminated area with it.
How is ammonium chloride used?
Many people don’t know, but the use of ammonium chloride will allow you to implement some tasks in such areas as:
- Mechanical engineering;
- Agriculture;
- Metallurgy;
- Forestry;
- Medicine;
- Chemical industry.
In addition, this substance can be used as a food additive "E510". It is mainly added to yeast, sauces and flour products. It should be noted that the use of ammonia as a food additive is prohibited in many EU countries. This is due to the fact that the substances included in its composition can cause harm to health.
How is hydrogen peroxide used?
A substance such as hydrogen peroxide is widely used in medicine and in agriculture. Thanks to its composition, the drug allows you to neutralize many diseases. Of course, it should be taken into account that it is used in conjunction with other drugs and substances.
If you don’t have bleach on hand, and you desperately need it, you can make it yourself. It is enough to add 200 milliliters of a 3% peroxide solution to the detergent.
Every modern girl dreams of having beautiful and smooth skin without the presence of irritating hairs. Most women still use a conventional machine, but few people know that Ammonia and Hydrogen Peroxide, which can be easily purchased at the pharmacy, can very easily solve this problem. This mixture was tested by applying it to copper. Also today there are many different ways to remove unwanted body hair.
Options such as:
- wax removal;
- sugaring;
- laser removal.
All modern newfangled methods have a lot of disadvantages:
- the procedures are generally quite painful;
- the effect after the procedure does not last long, which leads to the frequency of these operations;
- skin irritation almost always occurs;
- each method has its own side effects and contraindications;
- Most modern hair removal methods are only available by visiting specialists.
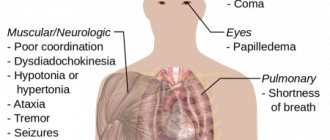
Classification of antiseptics
Halides (halogens and halogen-containing compounds) Chlorine or iodine compounds (antiformin, iodoform, iodinol, Lugol's solution, chloramine B, chlorhexidine).
The bactericidal effect is based on the fact that upon contact with organic substrates, these products release active halogens - chlorine and iodine, which destroy the proteins of microorganisms. Due to their high bactericidal activity, they are widely used both in medical institutions and at home. Oxidizing agents (hydrogen peroxide, potassium permanganate, hydroperite). In contact with tissues, they release active oxygen, which creates unfavorable conditions for the development of anaerobic and putrefactive microbes. They are used to a limited extent due to their moderate bactericidal activity and short shelf life. Acids (salicylic, boric). A shift in pH to the acidic side leads to denaturation of the protoplasmic protein of the bacterial cell. Salicylic acid has a weak antiseptic effect, and boric acid has a large number of side effects associated with toxicity. Currently, antiseptics for treating skin are practically not used as antiseptics. Alkalies (ammonia, sodium tetraborate). Currently, drugs are practically not used as antiseptics due to their low antiseptic activity. Aldehydes (formalin, lysoform). Penetrating inside the microbial cell, they come into contact with the amino groups of proteins, which leads to cell death. The same effect explains the strong irritant effect on human mucous membranes and skin. Currently, they are used more for disinfecting surfaces in medical institutions. Alcohols (ethyl). They dehydrate tissues and irreversibly coagulate the proteins of microorganisms. They are used quite widely and have a pronounced antiseptic effect. In 2006, the WHO declared alcohol-based hand sanitizers to be the gold standard for hand sanitizers. Cationic antiseptics (benzyldimethyl-myristoylamino-propylammonium). The active substance acts on the membranes of microorganisms, leading to their death. It has a very wide spectrum of antimicrobial action, stimulates the immune system, and accelerates the healing process of wounds. Widely used in surgery, obstetrics, gynecology, traumatology, burn therapy, otorhinolaryngology and other fields of medicine. Salts of heavy metals (preparations of mercury, silver, copper, zinc, lead). The antimicrobial effect is associated with blocking the sulfhydryl groups of microbial enzymes. Use is limited due to toxicity. Dyes (methylene blue, brilliant green, fucorcin). They are active against gram-positive bacteria and cocci. Methylene blue has a very weak antiseptic effect and is practically not used. Herbal antibacterial drugs (urzaline, calendula tincture, imanin and others). Weak antiseptic properties. Rarely used. All these substances have different degrees of activity, antimicrobial spectrum and toxicity. To understand how to choose the right antiseptic, you need to be guided by all these characteristics in accordance with the intended purpose: primary wound treatment, treatment of festering wounds, or treatment of damaged mucous membranes or intact skin/mucous membranes. When choosing which antiseptic to treat a particular wound, you must also follow the instructions in order to avoid side effects, as well as determine the dosage required in a particular case. Let's take a closer look at the most popular antiseptics.
What's the best way to remove unwanted hair?
As you know, perhydrol or hydrogen peroxide is a drug that is used as an antibacterial and antiseptic agent. And ammonia or ammonia is most often used to bring a person to his senses. It is mainly used in cases where it is necessary to urgently treat a bleeding wound. But, in addition, this product has been successfully used to remove and lighten unwanted hairs.
Comparison of addiction between hydrogen peroxide and ammonia
Like safety, addiction also involves many factors that must be considered when evaluating a drug.
So, the totality of the values of such parameters as “syndrome” for Hydrogen Peroxide is quite similar to the similar values for Ammonia. Withdrawal syndrome is a pathological condition that occurs after the cessation of intake of addictive or dependent substances into the body. And resistance is understood as initial immunity to a drug; in this it differs from addiction, when immunity to a drug develops over a certain period of time. The presence of resistance can only be stated if an attempt has been made to increase the dose of the drug to the maximum possible. At the same time, Hydrogen Peroxide has a fairly low “syndrome” value, just like Ammonia.
Why should you use peroxide?
This method of inhibiting the growth of unwanted hairs has several advantages.
These include the following:
- affordable price for the drug, which is significantly lower than the cost of all fashionable procedures in beauty salons;
- This method is time-tested, it is not the first generation that has been using it;
- the method is absolutely painless;
- after the procedure, ingrown, irritating hairs do not appear;
- by giving preference to the use of Ammonia and Hydrogen Peroxide, you may not be afraid of bacterial infection;
- in addition to hair removal, skin whitening also occurs, which makes the skin more beautiful and attractive;
- new hairs that will begin to grow on the areas of the skin that have undergone the procedure will be thinner and lighter, almost invisible;
- After several procedures, the hairs will completely turn pale and disappear, and the procedures can be stopped.
Hair removal on arms and legs
It often happens that ugly and dark hair grows on the skin of the legs or arms. This is especially unpleasant if the owner of such vegetation is a woman. While hair on the ankles can be shaved periodically, on the arms it is too much. Therefore, in such a difficult situation, a good solution would be to lighten the hair and, as a result, suppress the growth of unnecessary hairs using hydrogen peroxide and ammonia.
For this procedure you will need:
- a couple of ammonia capsules;
- 25 ml water;
- a teaspoon of baking soda;
- 25 ml hydrogen peroxide.
Peroxide is mixed in a container with water, and then soda and ammonia are added. You need to check yourself for allergies by applying a drop of solution to the crook of your elbow. If the test does not give results and the reaction does not occur, then you can safely apply the mixture to the area of skin with unnecessary hairs. Leave for about 60 minutes and wash off with running water and soap. If the skin areas are sensitive and tender, then lubricate them with baby cream before the procedure.
Facial hair removal
The so-called mustache in women is a real problem. They, of course, are not always noticeable, but sometimes they cause a lot of inconvenience. But there is no need to despair. A solution of Ammonia and Hydrogen Peroxide will also help here. You just need to wipe the area on your face where there are unwanted bristles or hairs with a solution of ammonia and perhydrol. Over time, the antennae will disappear.
Bleaching hair with hydrogen peroxide
The wonderful mixture mentioned above will help you forget forever about annoying hairs on your legs, face, stomach, and bikini area. Hair lightening with Hydrogen Peroxide and Ammonia or ammonia is gaining popularity every day, shifting expensive and unsafe procedures among cosmetologists from their pedestal.
If we talk about the hair on the head, then you can also use this mixture. When using the solution before washing your hair, a woman will soon become blonde, her hair will lighten, become softer and silkier. You should use this aqueous solution for hair on your head with extreme caution, without exceeding the concentration and without overexposing it to your hair. And it’s best to use proven cosmetics for your head, because no one wants to lose or spoil their hair in this place.
Sources:
Vidal : https://www.vidal.ru/drugs/hydrogen_peroxide__36359 GRLS : https://grls.rosminzdrav.ru/Grls_View_v2.aspx?routingGu >
Found a mistake? Select it and press Ctrl + Enter
| To main Add to favorites Feedback » title=”Write a letter”>Write a letter |
Articles by section
- Scientific research
- Near-scientific
- Technical solutions (without IT)
- IT, Internet
- Auto
- Video
- Law
- Health
- Money
- Ideas
- Books
- Deception
- Trips
- Technique
- Chemistry
- Electronics, electrical engineering
- Other
Articles by date (many are always current)
- 2022
- 2021
- 2020
- 2019
- 2018 December
- November
- October
- September
- August
- July
- June
- May
- April
- March
- February
- January
Tested for myself
- Hepatitis C cure
- Buying and renting an apartment
- Should you trust a woman in Russia?
- Detailed analysis of the family code
- Detailed analysis of the criminal code
- Detailed analysis of the housing code
- Hemorrhoids as a life destroyer
- Dental implantation
- To help a graduate student
- Diary of an Osteopathic Patient
- Borrow money or how I got into trouble
- White list of stores
Creation
- My programs
- My scientific articles
- My devices
- My accessories
- Unique items
- Interesting places in Russia
- Interesting places in Moscow
- Photo
- CMS: WordPress or Joomla?
Other sections
- To main
- Site files
- Site map (with materials)
- Feedback form
- Links and recommendations
- Sitemap (without materials)
Evergreen articles
Scoliosis A textbook for men Poisons Family Code Children Pregnancy and a man Loneliness Crisis Borrowed money Carbon monoxide Carbon dioxide Dating sites If a person choked Baptism Relieve anxiety Unemployment Inflation: mathematics Cheap flowers Loss of consciousness Missing man Mr. Freeman Fire extinguishers Wedding Computer help Mother's love Skid on ice Generation ideas Books by Alexander Nikonov MRI Euthanasia
Statistics
Users : 1 Articles : 1528 Content views : 5851824
Latest tags
omicron vvg png vvg png ls recharge donor donation gallbladder gaming industry China mt 2 omicron lighting disgust paracetamol pv 3 mb pv 3 mb pugvmb recheck plafond pugvmb development section snils tract
Most popular tags
usb windows exchange protection apartment purchase section auto section accessories section video section money section law section health section ideas section it section books section science section deception section pseudoscientific section devices section other section solution section technology section chemistry section electronics section website
Most Read Tags
guiformat harm codes section auto section accessories section video section money section law section health section ideas section it section books section science section deception section pseudoscientific section devices section other section travel section solution section technology section chemistry section electronics section Saiga 12c exp 01 030 speed emotions
Random tags
ac pwr loss restart c media halmacpi.dll https onecms sound blaster audigy platinum abscess activated carbon Akulychev water analysis knight SN exhibitions twins indexing Kalinov spring mounts for TV midlife crisis smoking methods thread offline shutting off water psyche registration homemade
| 09.03.2018 11:13 | |
| Save & Share | Cleaning brass to a shine (03/09/2018). → |
By mixing hydrogen peroxide 3% and pharmaceutical ammonia 10% in proportions 1:1 by volume, it is possible to obtain an extremely fast way to clean copper from oxides: by rubbing or soaking.
Work with gloves, of course. The complete reaction appears to be the dissolution of copper oxides with ammonia; using peroxide as a source of water and a reaction catalyst (it does not work without it). The reaction is a mixture of several simple reactions, such as the decomposition of peroxide into water and oxygen. CuO + 4NH3 + 2H2O2 → Cu(NH3)4(OH)2 + H2O + O2; Cu + 4NH3 + H2O2 → Cu(NH3)4(OH)2 - copper should not be kept in solution. It turns out that printed circuit boards are also etched using this method. Copper oxide should etch faster than copper, so damage should be minimal. Naturally, oxygen bubbles will physically disperse ammonia into the air—ventilation is required. The result is pure copper in a few seconds. This method is suitable for cleaning brass (destruction of copper/zinc in the alloy) - but then you still have to rub the brass with a steel wool in water to remove any remaining zinc. The flag for removing copper from the solution is the cessation of gas formation. In this case, the copper comes out clean and undamaged. If you change the proportions of ammonia:peroxide 1:2, problems with copper begin: it becomes covered with a certain blue substance; probably no longer diammine copper(II) hydroxide. Tags:
- ammonia
- stranded
- copper oxide
- peroxide
- hydrogen peroxide
- ideas section
- pseudo-scientific section
- section other
- solution section
- technology section
- chemistry section
- electronics section
- copper cleaning
Last news
- Real capacity of power banks (01/28/2022).
- Paracetamol overdose (01/25/2022).
- Russian games in the dark (01/25/2022).
- Hello, Omicron (01/24/2022).
- Battery discharge tester v.1.0 (01/22/2022).
- Test of Chinese resistors (01/20/2022).
- Blood donation (01/18/2022).
- Car battery - in a UPS (01/17/2022).
- Wire cross-section: deception (01/16/2022).
- Maintenance of the second ceiling chandelier (01/15/2022).
- Exchange. Return (01/10/2022).
- Residential wiring load tester v.1.1 (12/31/2021).
- Explosion of milk on the stove (12/25/2021).
- Exchange. Crooks (12/22/2021).
- PC PSU load tester v.1.0 (12/18/2021).
- TAT for a year (12/16/2021).
- Software announcement. Multimedia Keyboard (12/16/2021).
- Arduino and transients (12/15/2021).
- Exchange. Debts (12/15/2021).
- Opening of apartments and cars by the police (12/09/2021).
- Acidity of chewing gum (12/02/2021).
- About the Beep function (12/02/2021).
- Meter verification: overdue (11/16/2021).
- Rumble in the wall of the apartment (11/14/2021).
- Crown on a tooth (11/11/2021).
- About the machines in the dashboard (05.11.2021).
- Light switch replacement (11/04/2021).
- Replacement of passport. Consequences (28.10.2021).
- Bottles of aggressive medications (10/26/2021).
- Software announcement. Telemetry generator (10/25/2021).
- Maintenance of the ceiling chandelier (10/17/2021).
- From regular keyboard - multimedia (10/16/2021).
- Emotions at the end of the year (10/15/2021).
- Selecting days for vacation (10/15/2021).
- Debt to parents (10/14/2021).
- Pension. Mathematics (10/12/2021).
- Exchange. Honest casino (10/11/2021).
- Life in the Russian Federation is decay (09.10.2021).
- Hacking of bank accounts (09.10.2021).
- Exchange. Consequences (09.10.2021).
- Exchange. Hypnosis (09.10.2021).
- Exchange. Abstinence. Fear (06.10.2021).
- Repair of Daewoo Matiz: scam of 100k (04.10.2021).
- Jumper for UPS battery (04.10.2021).
- Air washer repair. Part 2 (04.10.2021).
- Gunners among specialists (04.10.2021).
- Exchange. Payment of taxes (20.09.2021).
- Exchange. Buying dollars (09.20.2021).
- Testing black caviar (09.20.2021).
- SMS sending module for Arduino (09/14/2021).
©2008-2022. All Rights Reserved. Developer - » title=”Sergey Belov”>Sergey Belov
.
The materials on the site are provided on an “as is” basis. The author does not bear any responsibility and does not guarantee the absence of incorrect information or errors. All responsibility for the use of materials lies entirely with the readers. Posting materials from this site on other sites is prohibited without indicating an active link to this source site (Civil Code of the Russian Federation: Article 1259, paragraph 1 + Article 1274, paragraphs 1-3). Many articles do not expire. It makes sense to watch both 2011 and even 2008. Site policy: write an article and then update it for many years. A VTB card has been opened for material support of the site: 5368 2902 0040 0838. Advertisers! Stop spamming me by email with offers to advertise on this site. I have never been a spammer/advertiser and never will be!
What to do about the unpleasant smell of shoes?
Do you have an unpleasant odor in your shoes? So it’s time to think about how to get rid of excessive sweating of the feet and how to handle shoes to avoid the growth of bacteria that contribute to the formation of odor. With the help of our tips, you can choose a way to eliminate the smell from new shoes and a means to remove it if the shoes are soaked with sweat.
Rough men's shoes and elegant women's ballet flats, cheap shoes and designer pumps can begin to emit an unpleasant odor. What to do if the shoes “go bad”? How to disinfect it and remove unpleasant foot odor? There are many means to combat this scourge; choose the one that suits you. But first, decide why this happened: the reason for the smell lies in the shoes themselves or in excessive sweating of the feet.
Iodine/povidone-iodine
Used in an alcohol solution (the so-called “tincture of iodine”) or in Lugol’s solution. Only the edges of the wound are treated with iodine so as not to cause soft tissue burns. The great advantage of iodine is its wide spectrum of antimicrobial activity: it kills all major pathogens and, with prolonged exposure, even spores - the most resistant forms of microorganisms. The use of large amounts of iodine is contraindicated in cases of hypersensitivity to it, hyperfunction of the thyroid gland, thyroid formations, dermatitis, and kidney diseases. Application to mucous membranes is not advisable, especially in children.
How to get rid of the unpleasant smell of sweat from your feet
Perhaps we should start with our feet. If your feet sweat excessively, you won't need any shoes. Therefore, you need to fight increased sweating. For these purposes, the pharmacy offers a lot of time-tested products:
- deodorizing foot cream with antifungal effect “Green Pharmacy”;
- antiperspirant with talc for sweaty feet “Green Pharmacy”;
- "Formagel" in the form of a colorless gel;
- “Teymurova paste”, popular in the last century;
- multicomponent ointment with antibacterial and antifungal effect “Nepotoff”;
- “Borozin” is a special powder poured into socks;
- antiperspirant "Drisol";
- preparations "Bellaspon", "Belloid", based on herbs.
How to remove unpleasant odor from new shoes
Yes, this happens too. Before you have time to rejoice at your new clothes, it turns out that the stylish new shoes smell terribly of chemicals and rubber. In this case, the following remedies will be effective and safe:
- a weak solution of potassium permanganate or hydrogen peroxide: treat the inside surface of new slippers or ballet shoes with them;
- vinegar and ammonia solution: soak a cotton pad in vinegar and put it in your shoes overnight; In the morning, wipe the inside of your shoes with ammonia;
- natural absorbents: pour flour, baking soda or talcum powder into problematic new shoes overnight, knock them out in the morning, and clean them with a vacuum cleaner;
- special chemicals: spray the inside of new shoes with shoe deodorant; Before putting on your shoes, wait until the product has completely dried.
Use of ammonia in medicine
In medicine, a 10% ammonia solution, known as ammonia, is widely used. Ammonia is used to revive an unconscious person. It is also used as an emetic in a very weak solution and in small quantities. Insect bites are treated with ammonia. Surgeons treat their hands with ammonia diluted in water.
Important!
Ammonia solution should be used with great care and only for its intended purpose. An overdose is very dangerous!
Prevention is the best way to combat bad shoe odor
By following a number of simple rules, you can not only avoid the appearance of a musty smell in your shoes, but also extend their life:
- Dry your shoes regularly: a humid environment promotes the growth of bacteria that causes an unpleasant odor. You can use regular newspapers to stuff your shoes with them, but special shoe dryers equipped with ultraviolet lamps will do the job best.
- Try not to walk for a long time in closed shoes, and when staying in a heated room most of the day, change your winter boots to shoes.
- Wear cotton socks that allow your feet to breathe.
- Take care of the insoles: dry them daily, wash them sometimes, change them every couple of months for new ones; buy insoles that have antiseptic properties.
- Activated carbon placed in leather or suede shoes overnight will not only absorb moisture, but also remove bad odors.
- The dirt that inevitably appears in fabric sneakers can be easily removed by regular washing using detergents.
It will be easier to follow these recommendations if you get several pairs of shoes for each season. This will allow you to devote enough time to drying and airing your shoes.
Sweat odor in shoes: practical ways to get rid of it
It's not just tightly closed sneakers that can cause suffocation. Sandals with thin straps, made from a certain type of leather, can also pretty much ruin your life. In case your shoes start to smell bad afterward, there are a number of products that have received good reviews about their effectiveness:
- Dufta Fit deodorant spray with double action: it helps remove the unpleasant odor of shoes and feet, acting on the source of the stench at the molecular level. The price of the drug depends on its volume.
- Treating shoes with chlorhexidine: a 0.05% solution of the drug is used for disinfection; Washed and brushed shoes are treated with a solution, also paying attention to the insoles on both sides, then the shoes are placed in a sealed plastic bag for four hours, after which the shoes are dried for a couple of days.
- Treating shoes with Formidron gives good results: it is better to carefully pour it into a bottle with a spray bottle and treat the inside of the boots. When working with the drug, you should be careful to avoid getting it into the respiratory tract.
- Special freshers are perfect for sneakers: convenient stickers, the antibacterial properties of which quickly neutralize the characteristic odor of sports shoes, and the design allows you to regulate the intensity of the action.
For those who prefer simpler home remedies for odor in shoes, ammonia, vodka or strong green tea will come to the rescue. You need to moisten your shoes with these products using a cotton pad, then dry them thoroughly.
When using any preparations, be careful, especially when processing light-colored, dyed and suede products. Make sure that there are no marks left on the outside of your shoes. Maintain personal hygiene, use the tips given, and you won’t have to blush when you take off your shoes at a party.